It implies that although processes tend to evolve naturally towards more significant disorder (higher entropy), they may be reversed if enough work is done to reduce that disorder again. This law has had a significant impact on science and technology. As the amount of energy in something increases, its thermodynamic entropy will go up until it reaches an absolute temperature before collapsing. (It may increase or stay the same.)įor example, when the ice melts, it increases the entropy of its environment because heat energy has become more random in its new liquid form. The concept of entropy is often associated with the second law of thermodynamics, which states that the entropy of an isolated system will never decrease over time. What Does Entropy Look Like in Science?Įntropy as a word is most commonly used in thermodynamics, quantum mechanics, statistical mechanics, cosmology, logarithms, and many other types of physics and information theories. Rudolf Clausius combined that with the Greek word tropē, which translates to “transformation.” It means things that transform within themselves due to their internal energy when translated literally. The prefix “en-” is commonly affixed to the beginning of words to show that the word is inside of something. However, just like many words, the word itself has its origins in the Greek language and prefixes from the English language. In 1865, German physicist Rudolf Clausius coined the term entropy during the development of his 2nd law of thermodynamics. What is the Etymology of the Word Entropy? When trying to get a better understanding of the entropy definition, it can be helpful to look at words with similar meanings here are some examples of entropy synonyms: This is a state of disorder at the highest degree and happens when the entropy increases to a place where the amount of heat is unsustainable within a closed system. 
Everything is working towards maximum entropy, which leads to what some scientists call the “heat death” of the universe. This is a non-reversible process that deals with a massive amount of free energy. This word is most commonly used within the bounds of the entropy of the universe. Still, even before something is old enough to be considered aged, it may have higher levels of disorder simply because it was produced in a disorderly way while being made (or assembled) by humans or nature. The natural process of aging can certainly take its toll on things. In other words, things fall apart as they get older until they reach total entropy. The second law of thermodynamics states that entropy in an isolated system increases over time, meaning that the conservation of energy becomes less functional over time and eventually will become useless. It is usually represented by the Greek symbol Θ (theta), which looks like an uppercase letter “V.” In thermodynamics processes, the definition of entropy is the degree of disorder in a system. It may be a complex concept, but the basics aren’t too much to handle! What Does Entropy Mean? Here is everything you absolutely need to know about entropy, where it comes from, and how to use it. However, learning the basics of entropia and its various meanings in the modern world can help to give you an edge in communication by using such complex words. Truly understanding entropic changes within a thermodynamic system can take years of high-level education in science. While that might seem simple enough, understanding entropic concepts can be confusing and complex. A more ordered state has less uncertainty and thus less entropy than a more disordered one. 
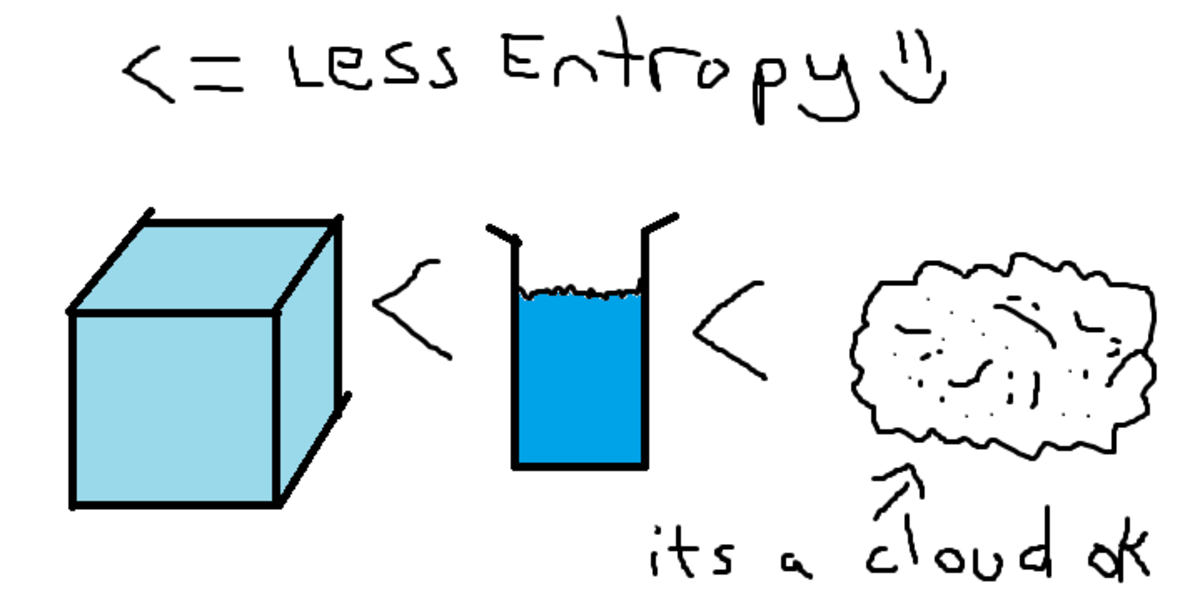
Entropy quantifies uncertainty and works as a measure of the disorder. In physics, entropy is a measure of how uncertain we are about the state of a system. 
Entropy is a concept that frames the evolution of systems from order to disorder, and it permeates everything around us. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
